Ensure Compliance with Document Control Software
Intelex Document Control Software simplifies document management across your organization, saving time and reducing errors. Centralize documents, speed up approvals and ensure security—all in one application.
Trusted by Leading Organizations Around the World





Efficient, Secure and Centralized Document Management
Centralize Document Control
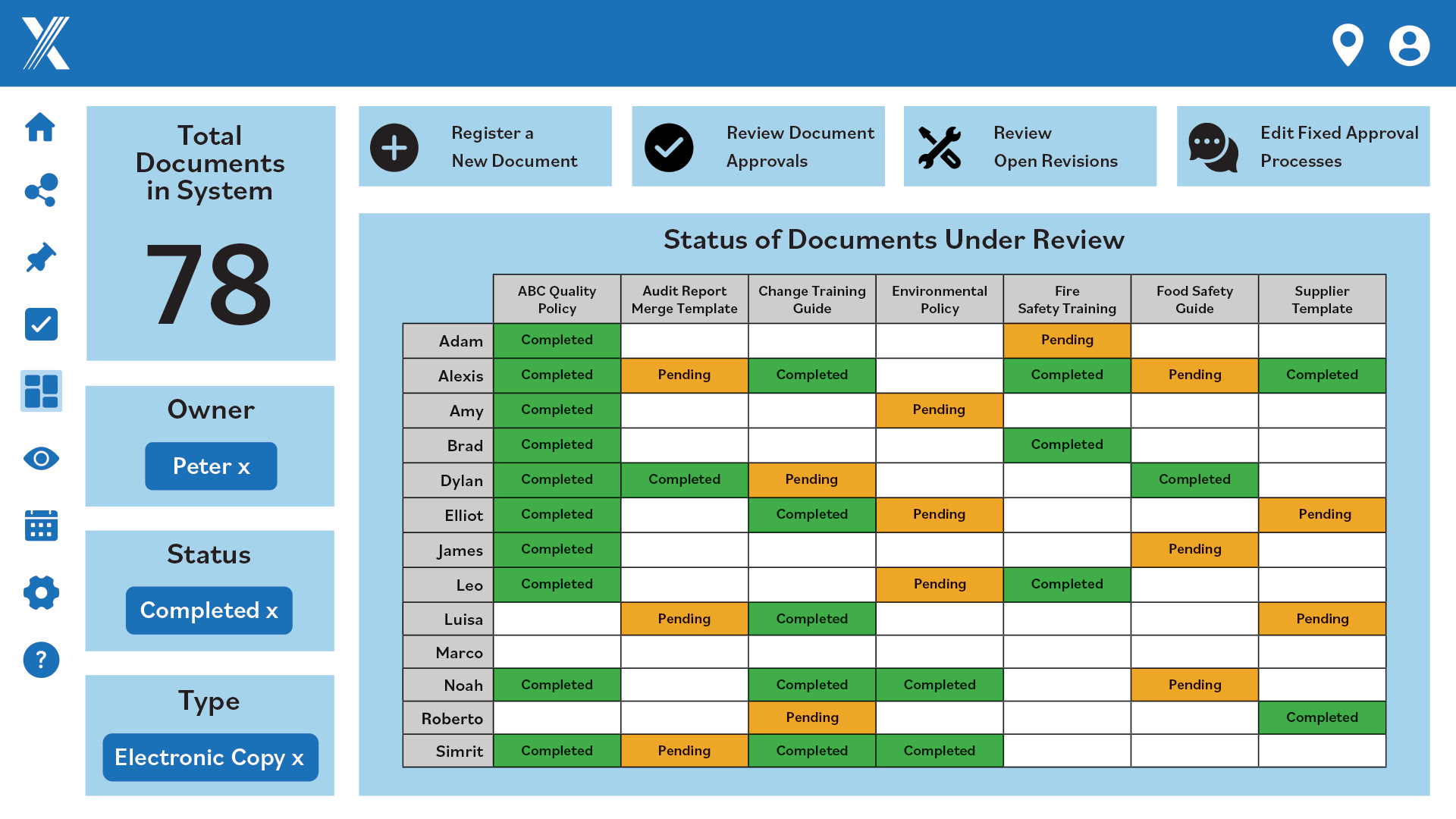
Disorganized documents and outdated information can lead to costly mistakes. Centralize documentation with automatically updated records and intelligent global search for easy access and efficiency.
Speed Up Reviews and Approvals
Manual reviews and approvals waste time and resources. Automate your document processes, streamline changes and ensure consistency with integrated lifecycle management and customizable templates.
Enhanced Data Privacy and Security
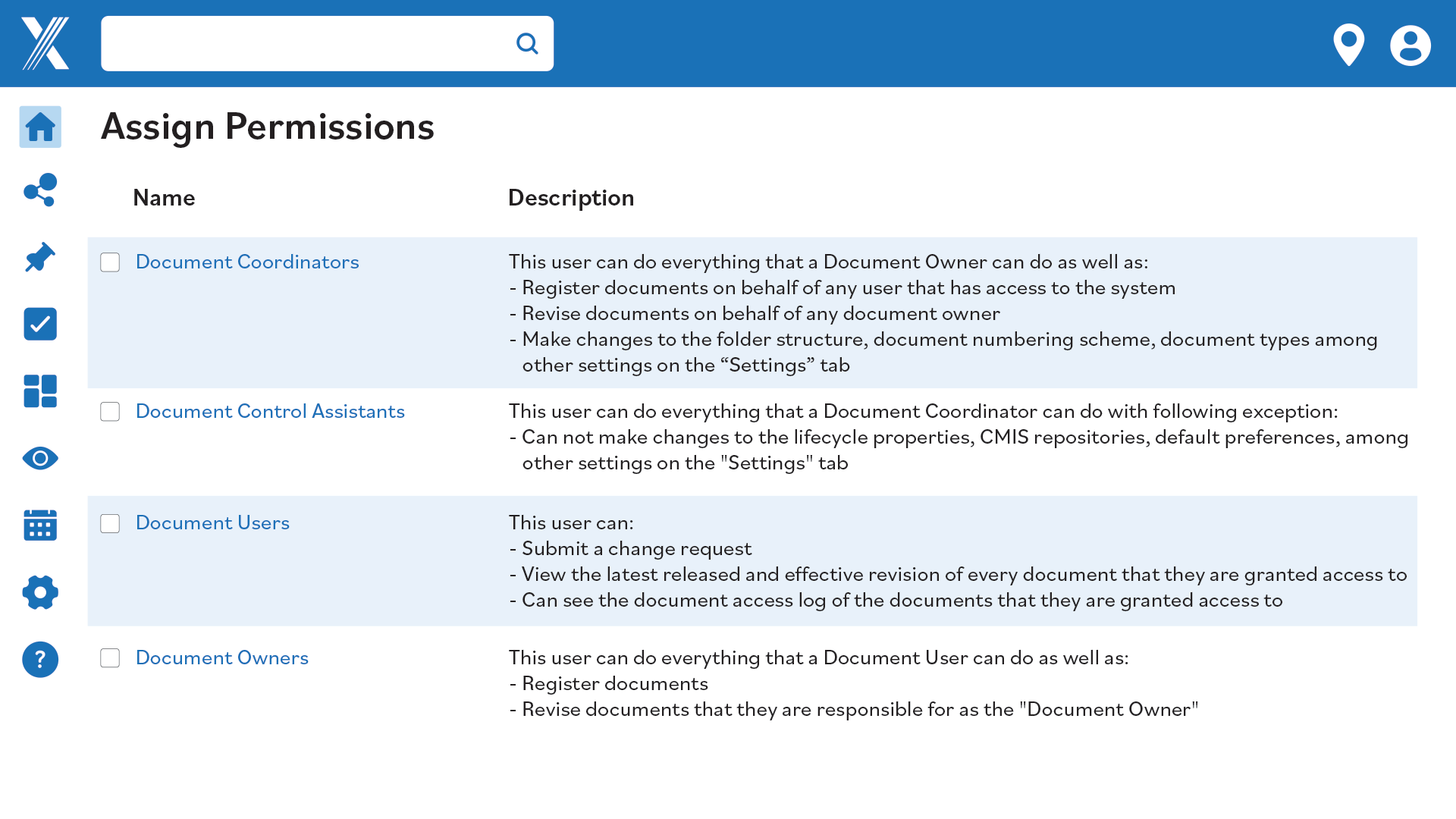
Sensitive documents are vulnerable to unauthorized access. Protect your data with document control software that offers robust access control and encryption.
Key Document Control Software Features
Intelex Document Control software offers powerful features to help you
customize and streamline your document management easily.

Folder Structure
Organize documents effortlessly with a customizable folder structure, ensuring easy access and efficient management.

Audit Trail
Track every document change with an audit trail, ensuring transparency, accountability and compliance.
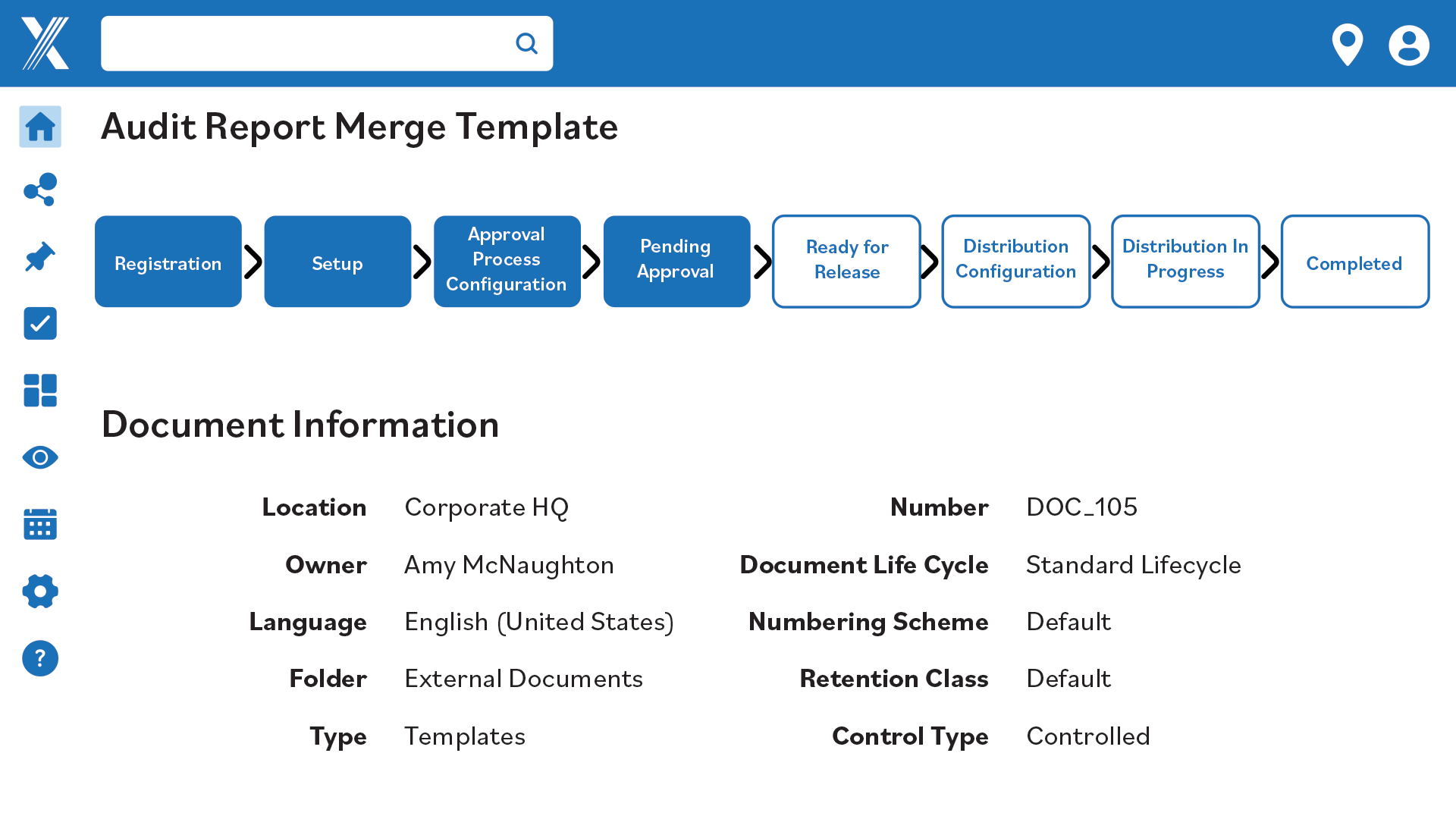
Revision Management
Manage document revisions by starting new versions, making old ones obsolete and tracking changes.

Workflow Status
Monitor document progress with workflow status, providing real-time updates on reviews, approvals and task completion.

Link Builder
Generate shareable public URLs, allowing easy document access without login.

Acknowledgement
Ensure compliance by having employees acknowledge they've read policies or completed trainings.
Streamline EHS Compliance for Supervisors
and Empower Frontline Workers
EHS supervisors streamline compliance, while frontline workers access up-to-date, secure documents with ease.

- Centralize compliance documents for easy management and access.
- Ensure audit readiness with automatic version tracking.
- Automate document approvals to reduce administrative tasks.
- Control access to sensitive EHS documents for enhanced security.
- Quickly access up-to-date documents when and where they need them.
- Reduce errors with easy access to the most recently approved procedures.
- Stay informed with real-time updates on document changes and revisions.
- Work confidently with secure access to essential EHS documents.
Document Control Software Frequently Asked Questions
Document control sets the information governance framework for how documents are approved, updated or amended, establishing that the organization is accessing accurate and reliable data and information. It is a critical aspect of risk management and supports standards like ISO 9001, ISO 14001, ISO 45001 and many others.
Document control software is a simple but powerful way to automate processes and workflows across the lifecycle of your company’s compliance efforts. Switching from a manual system to an automated process reduces human errors, saves countless hours of labor and allows your team to work more effectively on high-impact tasks.
Document management is the storing, locating, updating, tracking and sharing of documents. Both large and small businesses will use document management software, such as SharePoint or OpenText, to enhance productivity.
Document control shares all these features and functions, but also includes security, revision history and an approvals process. This makes it more appropriate for companies operating in regulated industries where compliance is mandatory or critical for success.
Document Control software saves time and money by automating key document management processes, preventing costly errors and centralizing access to vital documentation, such as forms, reports, schematics, plans, checklists and more.
- Centralize Documents: Organize documents within a single application with intelligent search capabilities that help you find the most recently updated data and information.
- Integrated Workflows: Allow collaborative edits of a single document with comments and side-by-side viewing. Once a document is approved, it is automatically updated to everyone with access.
- Enhanced Access Controls: Administrators can control the security of a document through each stage of its lifecycle. Security can also be modified depending on a user’s role and location.
- Maintain Compliance: Create a complete revision history so that all critical documents are ready for an audit at any time. Reduce the risk of penalties and easily adhere to compliance standards such as ISO 9001, ISO 14001, ISO 13485, and ISO 45001.
Regulated industries face unique challenges regarding risk management and compliance. They must manage high volumes of critical data, strict reporting requirements and frequently shifting compliance regulations. Document control not only helps these companies enhance productivity and prevent costly errors, but it is also an essential component of ISO 9001 and ISO 45001.
The most regulated industries are:
- Oil and gas extraction, processing, purifying and refining
- Power generation, transmission and distribution
- Motor vehicle manufacturing
- Pharmaceutical and medicine manufacturing
- Healthcare
Yes, document control is an important aspect of compliance for both EHS and Quality Management, particularly in highly regulated industries. A document control system organizes documents efficiently, automates approvals and reviews, streamlines workflows and ensures that key documents are secure. It is also a critical component of ISO 45001, ISO 14001, ISO 9001 and many other standards.
Document Control Software
Product Demo
Discover how our Document Control Software can benefit your organization. In this demo, see how to centralize document management, streamline workflows, ensure compliance and secure sensitive information. Learn how easy it is to automate approvals, track revisions and provide access to the latest documents.
Watch and learn how this application can:
- Centralize document management for streamlined workflows.
- Automate approvals and reduce manual tasks with ease.
- Ensure compliance with audit trails and version tracking.
- Secure sensitive documents with controlled access features.
Send the product demo my way!
"*" indicates required fields
 1 877 932 3747
1 877 932 3747









